Introduction
XRF (X-ray Fluorescence Spectrometry) is a chemical analysis tool commonly used for the quality control of raw materials and finished products in many industries including cement, mining and industrial minerals. The capabilities of XRF instrumentation have advanced significantly in the past decade in both sensitivity and stability such that they are no longer the most important source of error in an analysis. Sample preparation is now by far the most significant source of error in the analysis of materials by XRF. Viewed from this perspective, it is clear that high quality sample preparation techniques and practices are the key to achieving consistent and reliable analytical results.
One of the most common methods of preparing samples for XRF analysis is by making pressed pellets. This process is particularly popular as it produces high quality results, is relatively quick and is a low cost approach.
Sample Preparation for Pressed Pellets
The process of making pressed pellets for XRF analysis includes grinding the sample to a fine particle size, mixing it with a binder/ grinding aid in a grinding or mixing vessel, pouring the mixture into a pressing die and pressing the sample at a pressure of between 15 and 35T. The resulting pellet or tablet is then ready for analysis. While this is a common and relatively straightforward approach to preparing samples for XRF analysis, there are several important aspects that should be considered when designing a sample preparation protocol. These include the particle size of the sample, the choice of binder, the dilution ratio, the amount of pressure applied to the sample and the thickness of the final pellet. An additional consideration is sample contamination.
Particle Size
One of the key aspects of preparing pressed pellets is to make sure that the sample is ground to a particle size of <75µm, but <50µm is ideal. Depending on the sample type and volume, this can usually be accomplished with a few minutes of grinding with a typical ring and puck pulverizing mill. Small particle size is an important factor in producing pellets that provide the best analytical results because it effects how the sample will compress and bind together when pressed. More importantly, samples with larger or variable particle sizes can lead to heterogeneities in the sample. The sampling depth or critical escape depth for elements contained in a sample is energy dependent and, therefore, different for every element. Longer wavelength elements such as Na will have smaller escape depths than shorter wavelength elements such as Fe. This means that Na analysis is only sampling the first 10µm or so of a sample and thus more susceptible to sample heterogeneities on that scale.
Larger particle sizes can result from the presence of hard-to-grind phases in a sample resulting in variable grinding of the sample. The presence of larger particle sizes at the analysis surface of the sample can lead to analysis errors through the "shadow effect" where larger grains shadow the X-ray signal coming from the smaller grains at the surface of the pellet.
Binder
The sample binder is literally the glue that holds the sample together. The optimal binder is usually a cellulose/wax mixture that homogenizes with the sample during grinding and will recrystallize under pressure to bind the sample together. These binders are available under various commercial names but are generally similar. Sometimes acrylic binders are used, but these are difficult to homogenize with a sample in a mill and need to be mixed by hand. Some binder grinding agents are available as pre-mixed pellets that can be automatically added to a mill during grinding.
Dilution
The amount of binder added to a sample is an important consideration because it dilutes the sample and must also be added in the same proportion to every sample in order to avoid dilution errors. Since most modern XRF instruments provide substantial intensity for major elements it is safe to use a significant amount of binder in order to insure a good strong pellet. Weak pellets run the risk of breaking in the spectrometer potentially causing damage to the instrument. A 20-30% binder to sample ratio will almost always produce a very strong pellet that can be dropped onto the ground from a half a meter without breaking. If consumable costs are a concern, a lower binder/sample ratio can be used with some experimentation in order to determine the optimal dilution level.
Pressure
Once a sample and binder are mixed, the material is pressed in a die using a hydraulic sample press. The pressure applied to a sample should be sufficient to compress the sample completely and recrystallize the binder. One of the most important factors is making sure that the sample is fully compressed so that no void spaces remain in the pellet. A pellet may look good but still contain void spaces which can result in a lowered intensity for lighter elements. You should experiment with your samples using increasing pressure until the intensity for the light elements reaches a maximum and stabilizes. Most samples will reach this maximum at 25-35T of pressure applied for 1-2 minutes. It's also important to release the pressure applied to a sample slowly to prevent cracking of the sample surface.
Hydraulic Pellet Press

High quality pressed pellets ready for analysis
Thickness
The thickness of your pellet is important to achieve the best analytical results. The pellet must be infinitely thick to the X-ray beam for all of the elements that are being measured. X-rays produced in the sample must be able to escape the sample without being reabsorbed in order to make it to the detector to be measured. Higher energy elements (usually those with higher atomic numbers) will have deeper escape depths in a sample relative to lower energy elements. The signal coming from higher energy elements sample more of the pellet than those coming from lower energy elements. The sample's ability to reabsorb X-rays is directly proportional to the sample's average atomic mass. The absorption capability of any sample can be calculated from its average elemental composition and the mass absorption coefficient for each element present.
The problem arises when the escape depth of a particular element is larger than the thickness of the pellet. Because this is an element dependent issue, a particular pressed pellet may provide good results for some elements but not others. In general, you should make sure your pressed pellet is thicker than the escape depth of the highest energy element you want to measure.
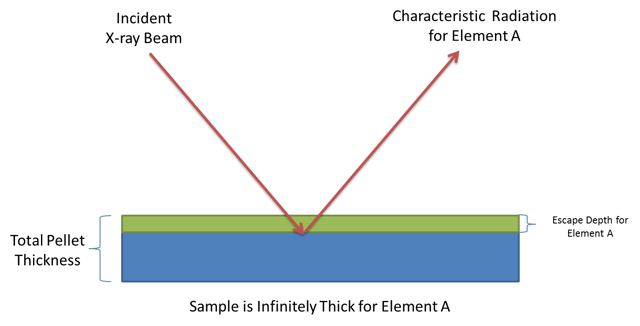
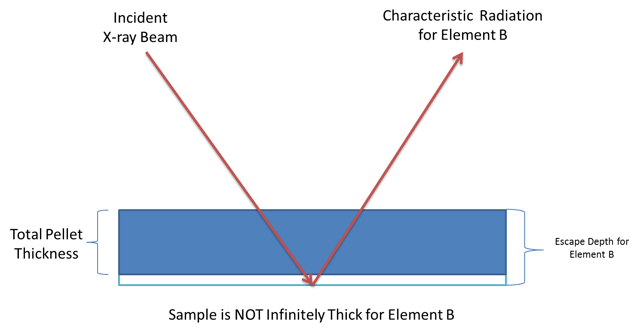
Usually a pellet made with 8-10g of sample for a 32mm diameter pellet or 13-15g of sample for 40mm diameter pellet will be sufficiently thick for the elements that can be measured by even the most powerful WDXRF instruments currently available.
Contamination
Sample contamination is an additional consideration in the preparation of high quality pressed pellets for XRF analysis. Contamination usually occurs during the sample grinding process and originates from two main sources: the sample preparation device and sample to sample cross contamination.
Contamination from the Sample Preparation Device
Sample pulverizers are the sample preparation devices that have the potential to contribute the most contamination to a sample. Pulverizers usually use a ring and puck style grinding bowl to grind samples from coarse chips to a fine powder. This grinding action can add various elements contained in the grinding vessel to your sample which is why care must be used in choosing your grinding medium. Steel, tungsten carbide and ceramics such as alumina and zirconia are the most common choices. Steel can add Fe, Ni and Cr, tungsten carbide will add W and alumina and zirconia will add Al and Zr respectively. It is, therefore, important to consider what elements are being analyzed and choose the grinding vessel accordingly to avoid contamination. Tungsten carbide is often a default choice because of its hardness and because W is usually not a key element for analysis in many applications. The downside is that tungsten carbide is often the most expensive option.



Alumina Grinding Bowl Tungsten Carbide Grining Bowl
Sample to Sample Cross Contamination
Sample to sample cross contamination in the sample preparation process is potentially the biggest overall contributor to sample contamination. This is particularly true when working in an environment where a wide range of sample types are being analyzed.
Preparation of Calibration Standards and Routine Samples
XRF is by definition a comparative elemental analysis technique where the instrument is calibrated with certified reference materials and unknown samples are measured against the resulting calibration curve for each element or oxide. It is therefore critical that all calibration standards and unknown samples be prepared in the same manor to ensure the accuracy of your analysis. Given this requirement you will need to develop a method that will address the issues detailed in this article for the full range of calibration materials and routine samples that you will need to analyze in your laboratory.
Summary
The most common contributors to error in the preparation of pressed pellets include the particle size of the sample, the choice of binder, the dilution ratio, the amount of pressure applied to the sample, the thickness of the final pellet and sample to sample cross contamination. The best practices for limiting this error require good method development coupled with attention to detail and consistency.
 We can provide a complete range sample preparation equipment and reference materials for XRF analysis. Need some help ? We have a team of specialists who have extensive experience with XRF as well as other analytical techniques. Our team can help you choose the right sample preparation method, reference material or provide some insight into your analytical challenges.
We can provide a complete range sample preparation equipment and reference materials for XRF analysis. Need some help ? We have a team of specialists who have extensive experience with XRF as well as other analytical techniques. Our team can help you choose the right sample preparation method, reference material or provide some insight into your analytical challenges.


